A Screening-Specific Disposable Endoscope (Gastroscope)
The World’s First Endoscopic System Designed Exclusively for Screening
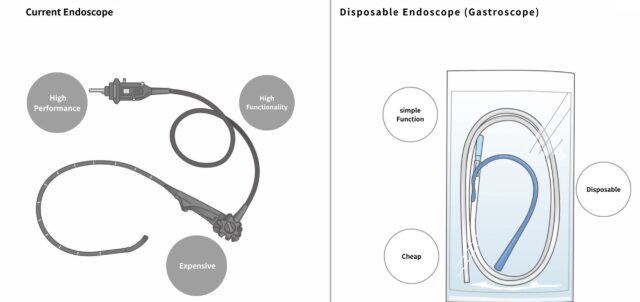
For more than 50 years, gastric cancer screening in Japan has been dominated by barium-based examinations. While a gradual shift toward endoscopic screening is now underway, existing endoscopes have evolved into highly sophisticated tools for diagnostic and therapeutic use. Their high cost—often several million yen per unit—and operational complexity make them unsuitable for large-scale screening.
Founder representative Kiyokazu Nakajima, a medical device development expert, recognized this structural issue early on. The requirements for diagnostic procedures and screening are fundamentally different. Screening primarily targets healthy individuals and requires efficient detection of suspicious lesions—not the advanced functionality needed for treatment.
Based on this insight, we adopted a “zero-based development” approach, building only the functions necessary for screening from the ground up. This has led to the development of the world’s first screening-specific disposable endoscope (gastroscope).
A New Screening System Enabled by Four Innovations
Ultra-Thin Design for Minimal Discomfort
Our device achieves the world’s thinnest diameter, optimized for transnasal insertion. This design significantly reduces gag reflex compared to conventional oral endoscopy. With simplified up-and-down controls, examinations can be performed efficiently with only the functions required for screening.
Fully Disposable, Zero Downtime
The most significant innovation is its fully single-use design.
By pricing the device within the typical out-of-pocket range of current gastroscopy screening, it can be adopted without increasing patient burden. The elimination of cleaning, disinfection, and drying processes allows examinations to be conducted continuously with zero downtime.
This dramatically increases throughput at screening centers while reducing operational costs.
In addition, the system connects directly to a tablet device, eliminating the need for expensive endoscopy towers. By integrating an LED light source at the tip, fiber optics and high-cost external light sources are no longer required—significantly reducing infrastructure investment.
Sterilized, Individually Packaged
Each device is sterilized and individually packaged, achieving a higher standard of safety than conventional disinfection-based reusable systems.
Reusable endoscopes require cleaning, disinfection, and drying after each use, and infection risk can never be fully eliminated. In contrast, our disposable endoscope is sterilized during manufacturing and provided in sealed individual packaging.
Each patient uses a brand-new device, effectively reducing infection risk to near zero.
AI-Powered Diagnostic Support: Accuracy and Efficiency
While the hardware is simplified, the software incorporates advanced technology.
Our AI-powered diagnostic support system, optimized for screening, automatically detects suspicious lesions and generates reports. We have demonstrated that sufficient diagnostic accuracy for screening can be achieved without the ultra-high resolution of conventional endoscopes.
Each device is assigned a unique ID during manufacturing, enabling a one-to-one linkage between the endoscope and the patient. This ensures precise data management, enables automated reporting to patients, referral institutions, and municipalities, and creates a foundation for future epidemiological research using large-scale data.
Transforming Gastric Cancer Screening in Japan
For Healthcare Providers
Endoscopic screening can be introduced without significant upfront investment. Smaller clinics will be able to offer screening services, greatly expanding access.
With zero downtime, the number of examinations per day can increase dramatically, while reducing the workload and costs associated with cleaning and disinfection.
For Patients
The ultra-thin, low-discomfort design significantly lowers the psychological barrier to screening.
With lower out-of-pocket costs and no radiation exposure or infection risk, safer and more accessible screening can be widely adopted.
For Society
By increasing screening participation rates (currently around 40%), our solution has the potential to unlock a massive market of tens of millions of people.
Earlier detection and treatment will improve outcomes while contributing to the reduction of overall healthcare costs.
Pathway to Global Expansion

Following validation in Japan, we plan to expand into Asian markets, including South Korea, Singapore, Thailand, and China.
In emerging markets where endoscopy is not yet widely available, our solution has the potential to become the “first endoscopy” platform. Its fully sterilized design also enables broader applications, including intraoperative use.
We are currently developing our first prototype in collaboration with manufacturing partners. Following iterative improvements, we aim for market launch around 2029 to early 2030, with regulatory approval processes already underway.
We are committed to realizing our mission: creating solutions from within the body’s lumen—“endoluminal.”
By transforming gastric cancer screening in Japan and expanding this successful model globally, Endoluminal Solutions Inc. aims to redefine the future of healthcare.

